Menopause Basics
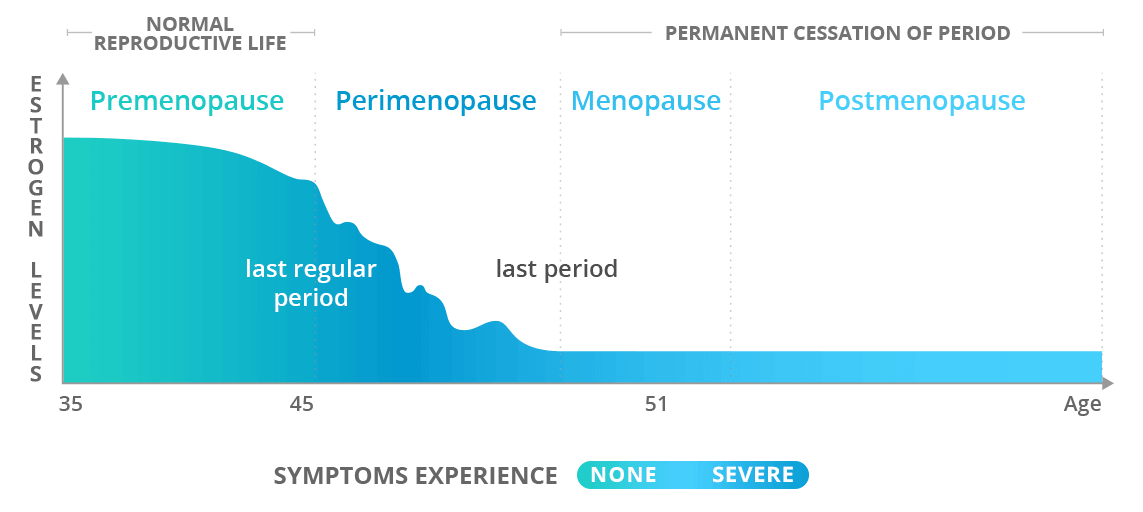
- Menopause is an inevitable phase in every woman’s life as her reproductive years come to an end.
- It is defined as the time when a woman has gone without a period for 12 months in a row.
- It occurs due to a gradual decline in reproductive hormones, estrogen and progesterone, because of natural aging.
- Menopause can also be induced through surgery, like hysterectomy, or certain medications.
- The average age of menopause is 51.
- Perimenopause, which can last from 2 to 10 years, is when most intense menopause symptoms occur, including irregular periods, hot flashes, and night sweats.
- A year after the last period, a woman is referred to as postmenopausal.
- Postmenopausal women should be cautious of complications such as heart disease and osteoporosis.
Causes of Menopause
Menopause happens as a result of a decline of hormones due to natural aging. As women age, the supply of their ovarian eggs, which are the main producers of key reproductive hormones, estrogen and progesterone, gradually decreases until it reaches total depletion. With no eggs being released, no hormonal production, and no periods occurring for 12 consecutive months, a woman is said to have entered natural menopause.